Updated by Faith Barbara Namagembe at 0955 EAT on Wednesday 6 2022.
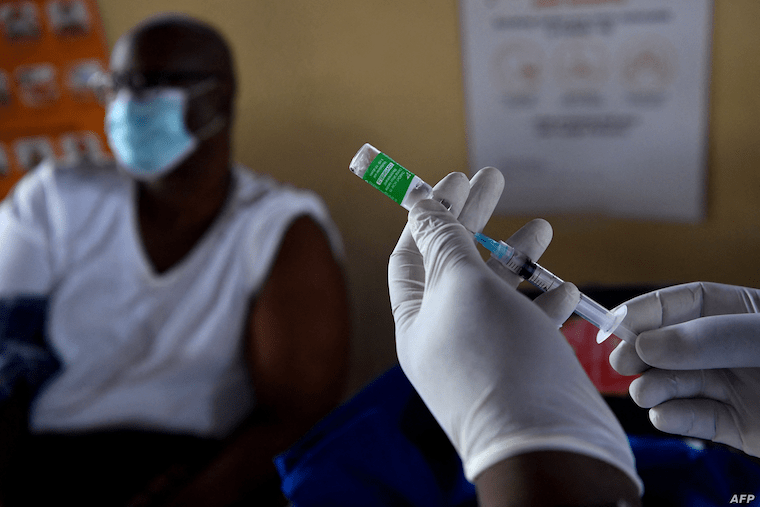
In 2021, Uganda launched the nation’s first local Covid-19 vaccine developing effort aimed largely at stepping-up the inoculation of as many Ugandans as possible.
Covid-19 infections and deaths remain low in Uganda while vaccination efforts are moving slowly that widespread immunity is unlikely in the country, raising the prospect of a bleak 2022.
As of April 1, 2022, the total cumulative Covid-19 cases were 163,936, new cases were four, active cases admitted at health facilities remain two, cumulative deaths 3,595 and total vaccine doses administered are19,095,668 out of a projected 30 million.
Globally, the pandemic accelerated the rush for the development of vaccines like Moderna, Johnson and Johnson, AstraZeneca, Sinovac, and over 170 vaccines under trials.
According to the Center for Disease Control and Prevention (CDC), Covid-19 vaccines are effective and lower risks of infection and transmission of the virus that causes Covid-19. Covid-19 vaccines also help in preventing serious illness and death in children and adults.
In 2021, Uganda embarked on developing its vaccine. Under the Presidential Scientific Initiative on Epidemics (PRESIDE), a brainchild of President Yoweri Museveni led by Dr. Monica Musenero, a presidential advisor on Epidemics, government set aside Shs 31 billion for the development of the country’s first-ever Covid-19 vaccine.
Scientists were supposed to conduct preclinical tests last month using model animals like mice and chimpanzees to determine the vaccine’s safety, immunogenicity and efficacy.
The preclinical tests are yet to kick off. Musenero, the minister for Science, Technology and Innovation has not explained the cause of the delay. Interviewed for an update on the development and manufacturing of Uganda’s Covid-19 vaccine, Dr. Musenero referred us to her personal assistance.
We couldn’t reach the assistant. Her phone was off. According to a source in the Regulation and Prequalification Department of the World Health Organization (WHO) in Geneva, Uganda is far from manufacturing a globally recognized Covid-19 drug/vaccine.
“This is not about writing off Uganda but they didn’t prepare before embarking on the project. The development and manufacturing of Covid-19 drugs/ vaccines is a fulltime job, which requires a team of professionals and modern equipment from the time of research to production,” he said.
“Uganda has a good idea and is on the right track and therefore, needs to be supported. But its drug monitoring body, the National Drug Authority (NDA), is underfunded and, therefore, or unless otherwise cannot supervise the process,” the source said adding that, “there is no recognized drug monitoring body in the region as far as the development of Covid-19 vaccines is concerned except the South African Health Products Regulatory Authority (SAHPRA),” he said.
The source said the NDA will be required to collaborate with recognized regional bodies like SAHPRA to monitor the process when it comes to testing the product on human beings.
In February 2022, Dr Musenero, said that her team was facing a number of unprecedented challenges such as accessing key equipment in the development of vaccines.
“You work and reach a point where you need equipment; so, you have to stop the experiment and wait. We have also had a challenge of the reluctance of some suppliers of substances required in the process of developing the vaccine,” she said.
With the continuous pauses and lack of critical equipment, the country will wait for years to have its first vaccine tested and approved by WHO for use on human beings.
Currently, there are countries in Africa that produce vaccines. Senegal produces the yellow fever vaccine. There are ongoing negotiations and collaborations with advanced manufacturers of Covid-19 vaccines to transfer technology and patent rights. So far South Africa is the only country which has an end-to-end technology which produces Covid-19 vaccine. South Africa is producing Pfizer.
Abiaz Rwamwiri, the public relations manager of NDA, dismissed claims that the authority has no capacity to supervise the process of manufacturing Covid-19 vaccines.
“Those allegations are baseless, WHO is our partner and should have told us. I have not seen the official statement,” he said.
“Uganda’s scientists working on the process of manufacturing Covid-19 vaccine will need us when they are done with clinical trials in animals. They will need us when they want to test their vaccines on human beings,” he said.

